Food Safety Management System
The Food safety management system is based on the Special Act on Imported Food Safety Control(Special Act), which aims to contribute to sound order in trade and to improving people’s health by ensuring the safety of imported food, etc. promoting improvement in the quality thereof, and providing proper information.
- Safety control before importation: On-site inspections of foreign food facilities or establishments, Sanitation assessment of livestock products and products under special sanitary control*, Arrangements on sanitary control of imported fishery products *fish heads and edible part thereof, offal of edible fish and molluscs
- Safety control in customs clearance: Border inspection(documents review, field inspection, close inspection, random sampling), Orders to undergo inspection
- Safety control in distribution(domestic supply chain): Collection and inspection of imported food products, Inspection of overseas direct purchase of food products
| Safety control for the entire cycle from importation to distribution |
|---|
|
|
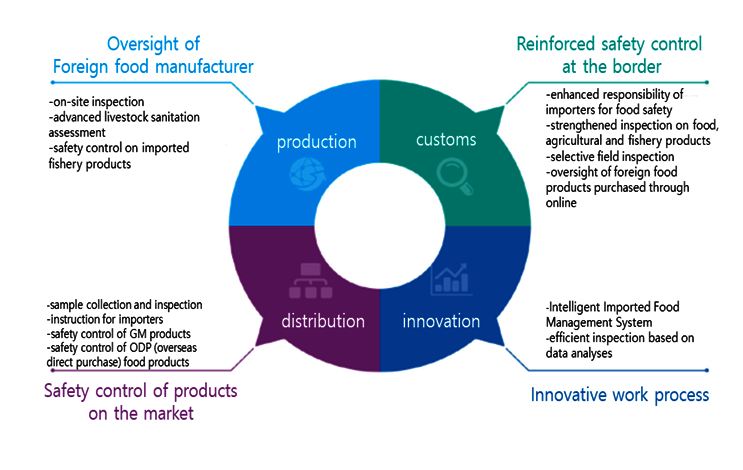
- production
- Oversight of Foreign food manufacturer
- on-site inspection
- advanced livestock sanitation assessment
- safety control on imported fishery products
- customs
- Reinforced safety control at the border
- enhanced reponsiblity of importers for food safety
- strenghened inspection on food, agricultural and fishery products
- selective field inspection
- oversight of foreign food
- products purchased through online
- distribution
- Safety control of products on the market
- sample collection and inspection
- instruction for importers
- safety control of GM products
- safety control of ODP(overseas direct purchase) food products
- innovation
- Innovative work process
- Intelligent Imported Food Management System
- efficient inspection based on data analyses
Relevant Laws and Regulations
The Special Act on Imported Food Safety Control
Notices of MFDS
- Inspections for imported food etc.
- Inspections for imported health functional food
- Import declaration and inspection of imported livestock products
- Countries allowed for livestock exportation & Import sanitation requirements
- Provision for food products subject to inspection order
- Training and designation & operation of training institution for business entities
- Procedure and standards for registration and on-site inspection of foreign food facilities
- Standard for registration and management of Good Importers
- Regulations for the works of imported food sanitation audit institution
- Standards for setting the expiration date of food, food additives, livestock products and health functional food
- Traceability of Imported Food
- Standards and specifications for food
- Standards and specifications for food additives
- Standards and specifications for health functional food
- Regulation on approval of functional ingredient for health functional food
- Standards and specifications for food additives
- Standards and specifications for food utensils, containers and packages
| Notices for on-site inspection, sanitation requirements, and inspections in the customs clearance |
|---|
|
|
| Regulations related to the standards and specifications for Imported food |
|---|
|
|
- Items for Import Declaration
- Imported food products etc.* for sales or business purpose ※ processed products, health functional food products, agricultural or forestry products, livestock products, fishery products, utensils, containers and packages
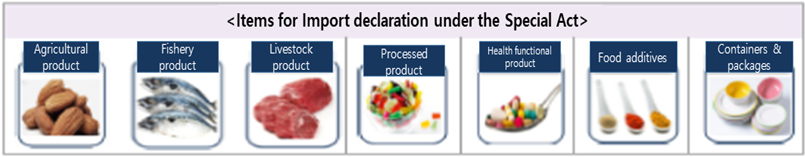
- Items for Import declaration under the Special Act
- Agricultural product
- Fishery product
- Livestock product
- Processed product
- Health functional product
- Food additives
- Containers and packages
- Food : All kinds of foods and beverages (except for foods and beverages taken as medicine)
- Food Additives : materials added to or mixed with foods or materials used for wetting foods in the process of manufacturing, processing or preserving
- Food utensils, containers and packages: machines or utensils used for eating or containing food, which come into direct contact with foods or food additives
|
|
- Health Functional Food : food that are manufactured or processed in order to supplement nutrients that may be deficient in daily meal or to provide functional ingredients useful for human body
|
|
- Livestock products : meat, packaged meat, raw milk, shell egg, processed meat products, dairy products and processed egg products
- (Meat) carcass, dressed meat, internal organs, and other parts of livestock for human consumption
- (Packaged meat) no additive of chemical compound or other food is added, as meat refrigerated or frozen as packaged, cut meat (including finely cut or grinding) for the purpose of sale
- (Raw milk) cow's milk or sheep's milk that has been milked for sale or for treatment and processing for sale
- (Shell egg) eggs of livestock used for food, prescribed by Ordinance of the Prime Minister
- (Processed meat product) products for sale prescribed by Presidential Decree, such as ham, sausage, bacon, dried meat, spiced meat, and other processed products with meat as a raw material * Ground meat product, rib product, meat extract, beef tallow, lard
- (Dairy product) products for sale prescribed by Presidential Decree, such as milk, low-fat milk, powdered milk, milk formula, fermented milk, butter, cheese, and other processed products with raw milk as raw material * Low-fat milk, lactose degraded milk, processed milk, goat milk, butter milk, concentrated milk, milk cream, wheys, hydrolyzed milk protein products, ice creams, ice cream powder types, ice cream mixes
- (Processed egg product) products for sale prescribed by Presidential Decree, such as liquid egg yolk, liquid egg white, whole egg powder, and other processed products with eggs as raw material * Whole egg liquid, yolk powder, egg white powder, heat-formed products, salted egg, pidan
|
|
Proactive safety control measures for imported food
In a regard of proactive safety control for imported food, registration to MFDS is a mandatory requirement for any foreign food facilities exporting their products to Korea and MFDS on-site inspection is conducted to verify sanitation status of foreign food facilities and to certify eligible establishments as a ‘Good Foreign Food Facility’.
Registration of foreign food facilities
- The registration as a foreign food facility to MFDS must be completed before import declaration.
- (Subject to registration) : Importers or business operators of foreign food facilities
- (How to register) : Submit online application through the Imported Food Information Maru (https://impfood.mfds.go.kr)
*Documents for submission: Either Letter of Consent for registration or an official government document proving business registration (in English) - (Processing period) : 3 days

- Registration procedure
- Application
- Receipt of documents
- Review
- Approval
- Notification of registration
Registration of foreign establishments
- Any person who establishes and operates a foreign establishment must file an application for registration with MFDS via the government of an exporting country before import declaration of livestock products.
- Documents for submission
- Application for registration(change in registration)
- A checklist to demonstrate regular inspection∙supervision conducted by the government of the exporting country in compliance with the inspection standards/criteria which the Minister of Food and Drug Safety establishes and provides.
- A copy of business approval(registration) documents issued by the government of exporting country
- In the case of a foreign establishments adopting own standards equivalent to standards for certification of sanitary control specified in 「Livestock Products Sanitary Control Act」, a summary of business operating plan and a copy of process flow chart with critical control points indicated.
- For an establishment not applicable to ④ above, summary of sanitary control standard the establishment complies with and a copy of process flow chart.
On-site inspection for foreign food facilities
- A person who intends to import food, etc. into the Republic of Korea or person who establishes and operates a foreign food facility shall register matters prescribed by Ordinance of the Prime Minister (refer to Article 5 of the Special Act on Imported Food Safety Control.
- If a foreign food facility has previous history of exporting non-compliant products or it is necessary to confirm facts about safety information such as hazards on food, on-site inspection is conducted through prior consultation with either the government of exporting country or the foreign food facility. * Applicable establishments should respond to MFDS on whether to accept the on-site inspection within 15 days after receiving MFDS notification and 20 days after MFDS sends the notification.
- If the government of an exporting country or a foreign food facility refuses, interrupts or avoids the on-site inspection including the case no response is made without any lawful reasons or if there is a concern that a hazard is likely to occur in imported food, MFDS will suspend the importation of the food products of the foreign food facility concerned.
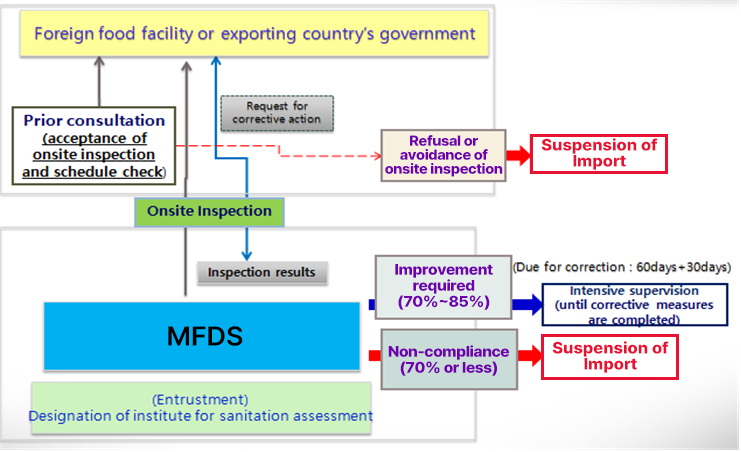 [The following diagram is about the description regarding on-site inspection]
First of all, foreign food facilities that want to export products to Korea shall register their manufacturing facilities for those products.
If a foreign food facility has a previous history of exporting non-compliant products or it is necessary to confirm facts about food safety information such as various hazards posed on food safety,
on-site inspection is conducted.
Prior to on-site inspection, a consultation with either the government of an exporting country or the foreign food facility is held to determine whether to accept onsite-inspection and to schedule on-site inspection.
Second, MFDS will conduct an on-site inspection of foreign food facilities according to the schedule consulted.
MFDS will suspend import if an on-site inspection is interrupted, refused, or avoided for any reason including the case no response is made without any lawful reasons or
if there is a risk that a food safety hazard is likely to occur in imported food of the foreign food facility concerned.
Once the inspection is completed, MFDS will share the results : if the compliance rate is 70-85% (requires improvement), a correction period of 60 days + 30 days will be given to work on corrective action,
and there will be intensive supervision until corrective action is completed. In addition, if the compliance rate is 70% or less, import is prohibited considering it as inappropriate.
[The following diagram is about the description regarding on-site inspection]
First of all, foreign food facilities that want to export products to Korea shall register their manufacturing facilities for those products.
If a foreign food facility has a previous history of exporting non-compliant products or it is necessary to confirm facts about food safety information such as various hazards posed on food safety,
on-site inspection is conducted.
Prior to on-site inspection, a consultation with either the government of an exporting country or the foreign food facility is held to determine whether to accept onsite-inspection and to schedule on-site inspection.
Second, MFDS will conduct an on-site inspection of foreign food facilities according to the schedule consulted.
MFDS will suspend import if an on-site inspection is interrupted, refused, or avoided for any reason including the case no response is made without any lawful reasons or
if there is a risk that a food safety hazard is likely to occur in imported food of the foreign food facility concerned.
Once the inspection is completed, MFDS will share the results : if the compliance rate is 70-85% (requires improvement), a correction period of 60 days + 30 days will be given to work on corrective action,
and there will be intensive supervision until corrective action is completed. In addition, if the compliance rate is 70% or less, import is prohibited considering it as inappropriate.
Import sanitation assessment for livestock products and food subject to special sanitation control and overseas on-site inspection for foreign establishments(food facilities)
- Import sanitation assessment system for livestock products and food subject to special sanitation control, which enables import approval by country, has been operated and if the sanitary control system of the exporting country deems equivalent to that of Korea resulting from total 6 steps of import sanitation assessment, products from the country is allowed for import. * 6 steps of import sanitation assessment (step 1) send import sanitation questionnaire to the exporting country → (step 2) review on answers of the questionnaire → (step 3) overseas on-site inspection → (step 4) determination of import approval → (step 5) consultation with the exporting country on import sanitation requirements and export health certificate → (step 6) registration of foreign food facilities and establishments
- Only the livestock products and food subject to special sanitation control produced by registered foreign establishments(food facilities) are allowed for import to the Korean market. Besides, the foreign establishments(food facilities) already registered are subject to on-site inspection conducted for the purpose of verifying the government’s sanitary control system of the exporting country and sanitation of individual establishments(food facilities).
Registration of ‘Good Foreign Food Facility’
- If an operator of a foreign food facility inspects and controls sanitation of the facility in a proactive way, after verifying the fact through on-site inspection, MFDS registers and manages the food facility as a ‘Good Foreign Food Facility’. * Subject to registration: name and address of the facility, name of business operator, exported products and other relevant information ** Relevant items: food and food additives, utensils, containers and packages, health food and health functional food
Activities for customs clearance safety control
Import declaration and inspection
- Where a business entity intends to import (including filing an import declaration by proxy) food, etc. for the purpose of sale or for the purpose of using them for business, he/she shall file an import declaration of the relevant imported food, etc. with the Minister of Food and Drug Safety.
- In accordance with the relevant regulations, food products etc. which filed an import declaration for the first time will be subject to a close inspection. The same products from the same company will be allowed for customs clearance only when they are found to be compliant after document review, field inspection, close inspection and random sampling.
- Especially, by using ’Intelligent Imported Food Management System‘, inspections could be enhanced on the basis of the results of statistical analysis of hazard information (i.e, country, product item, history of hazardous substances detection and overseas hazard alert). Also, with the Machine Learning technology, the efficiency of safety control of imported food could also be strengthened by selecting facilities or import items to be subject to on-site inspection, customs inspection or market inspection.
Orders to Undergo Inspection
- It is to make importers more responsible on the safety of food imports by requiring them to undergo inspection by an inspection agency prescribed by the Act on Testing and Inspection in the Food and Drug Industry before declaring importation when the products have a history of non-compliant or a high level of hazard.
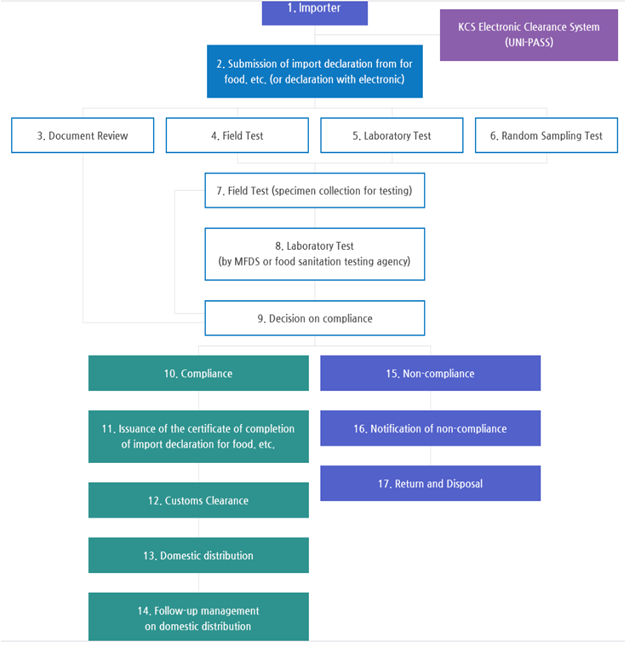
- 1. Importer
- KCS Electronic Clearance System(UNI-PASS)
- 2. Submission of import declaration from for food. etc.(or declaration with electronic)
- 3. Document Review
- 4. Field Test
- 5. Laboratory Test
- 6. Random Sampling Test
- 7. Field Test(specimen collection for testing)
- 8. Laboratory Test(by MFDS or food sanitation testing agency)
- 9. Decision on compliance
- 10. Compliance
- 11. Issuance of the certificate of completion of import declaration for food. etc.
- 12. Customs Clearance
- 13. Domestic distribution
- 14. Follow-up management on domestic distribution
- 15. Non-compliance
- 16. Notification of non-compliance
- 17. Return and Disposal
Activities for safety control in distribution
Collection & inspection, Instruction & Supervision
- The imported foods that are most frequently consumed on the market or those with non-complaint history, or those on the risk alert and with the potential food safety issues will be collected and inspected. In addition, the instruction and supervision will be given to the importers with history of administrative disposition to prevent illegal activities.
- When products found to be non-compliant or recalled, prompt discard/disposal will be carried out so that the safety of imported food products on the market can be ensured.
Safety control for overseas direct purchase of food
- With a growing consumption of food products directly purchased from overseas due to increased use of internet and social media, collection and inspection for those products are implemented. In line with this, MFDS warns customers not to buy such products online using publicizing methods.
- After conducting inspections on food products purchased directly from online and confirming the presence of any harmful substances, MFDS requests a withholding of the products concerned to the Korea Customs Service and publishing the list of such products on the website “FoodSafetyKorea”.

