About GHC
- The Global Harmonization Center (GHC) is an international training institution dedicated to regulatory harmonization.
- Established in January 2025 under the Ministry of Food and Drug Safety (MFDS) of the Republic of Korea, GHC plans and operates training programs to strengthen regulatory capacity in the field of medicinal products.
- Building on the accomplishments of the former APEC Harmonization Center (AHC), GHC collaborates with regulatory authorities, industry, academia, and international organizations to contribute to global regulatory convergence.
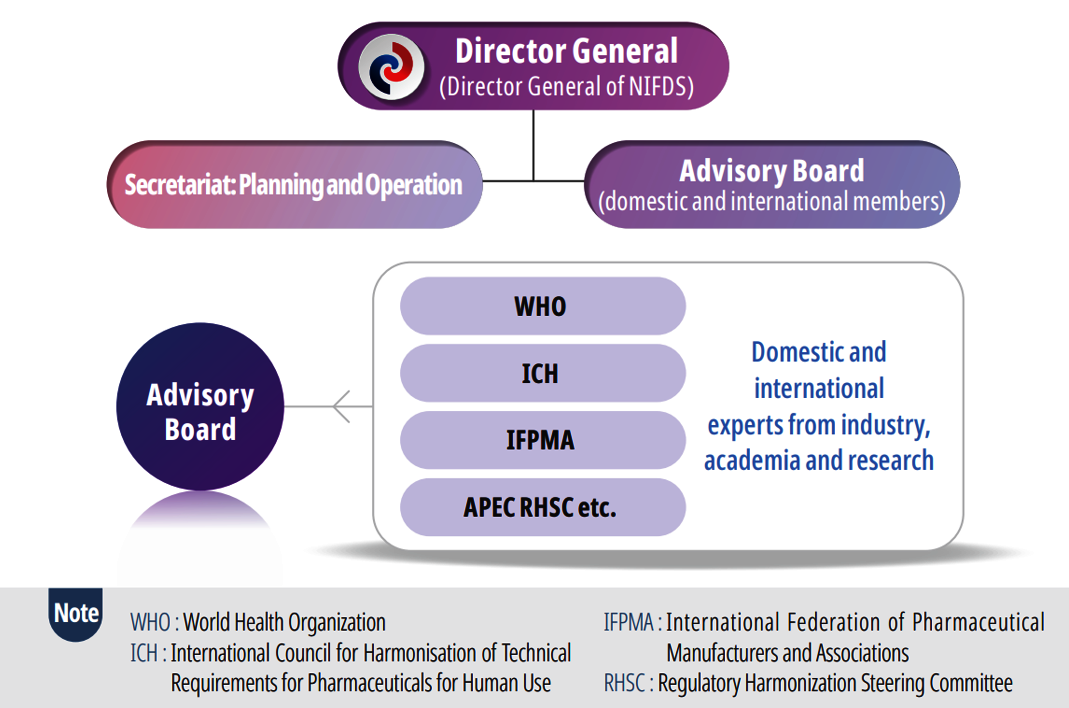
Organization
- GHC operates as the secretariat within the Pre-submission & Consultation Division of the National Institute of Food and Drug Safety Evaluation (NIFDS), MFDS.
- The secretariat is supported by an advisory group composed of domestic and international regulatory experts who provide guidance on the development of training content and strategic planning.
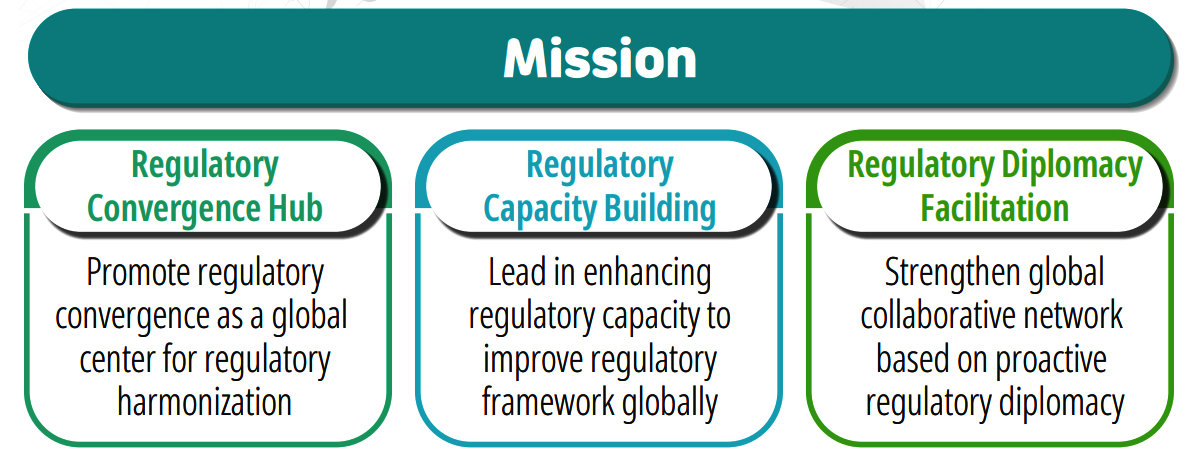
Mission
- The mission of GHC is to advance international regulatory convergence by promoting regulatory diplomacy and enhancing the capacity of regulatory authorities through structured education and cooperation.
- Through these efforts, GHC seeks to contribute to the improvement of the quality, safety, and accessibility of medicinal products worldwide.
Key Activities
- GHC carries out a wide range of initiatives to advance international regulatory harmonization and capacity building in the field of medicinal products.
- To fulfill its mission, GHC undertakes the following key activities:
1) Implementation of diverse training opportunities
- Delivering training programs in various formats—including workshops, hands-on training, webinars, and online courses via the GHC e-learning center, in collaboration with Korean and international regulatory partners (e.g., ICH, IFPMA, WHO, IVI, GTH-B, K‑NIBRT).

2) Dissemination of educational materials and regulatory information
- Sharing training resources and regulatory updates through the GHC official website and e-learning center
GHC Official Website
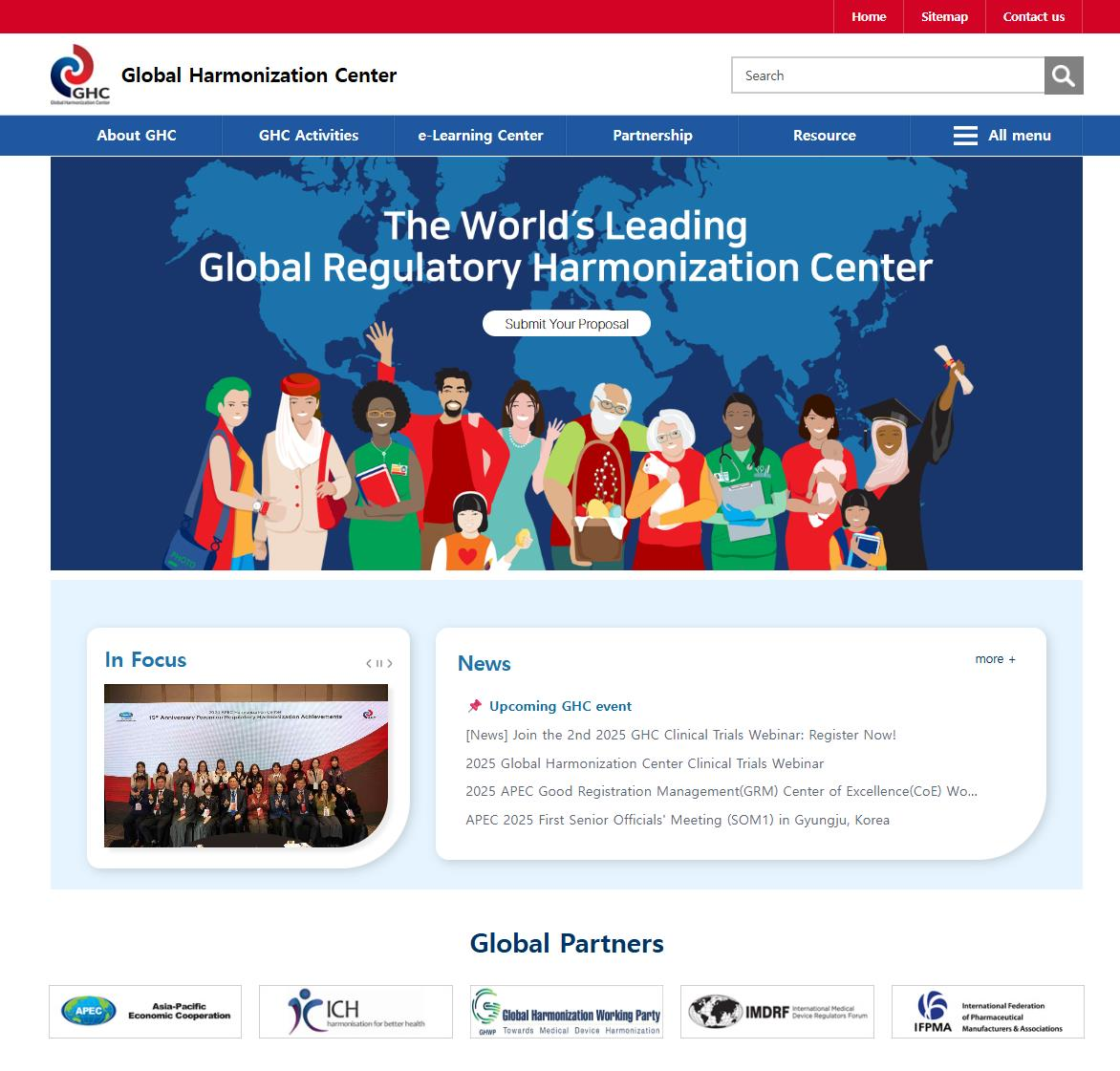
: The official platform of the Global Harmonization Center (GHC), providing comprehensive information on its key initiatives, training programs, and educational resources.

GHC e-Learning Center
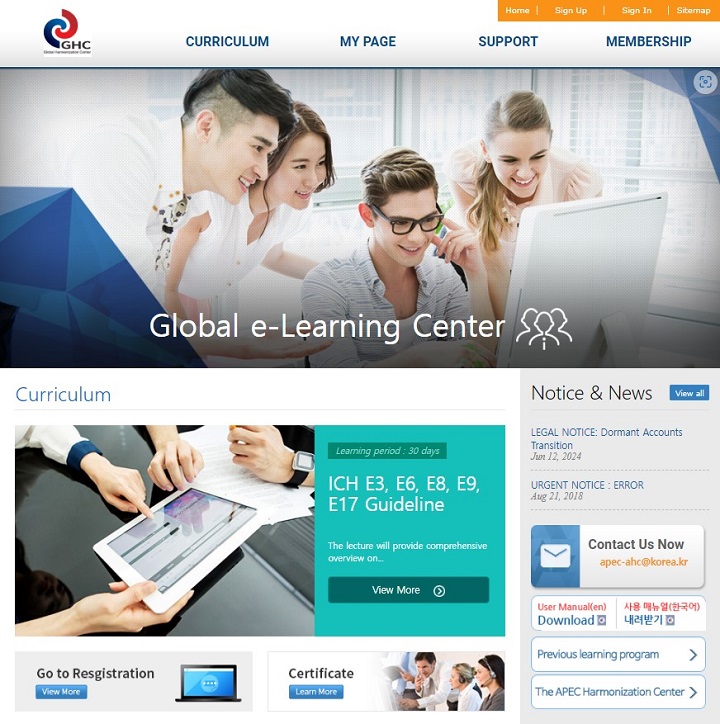
(https://www.nifds.go.kr/ghc/edu)
: An online learning platform offering introductory courses on ICH guidelines and other regulatory topics, with certificates awarded upon successful completion.





